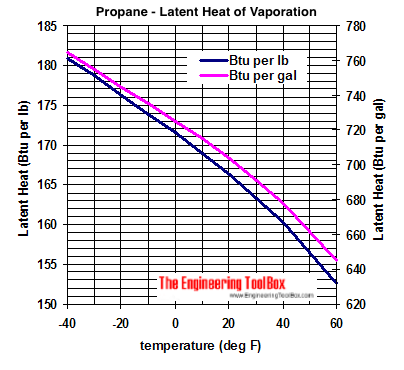
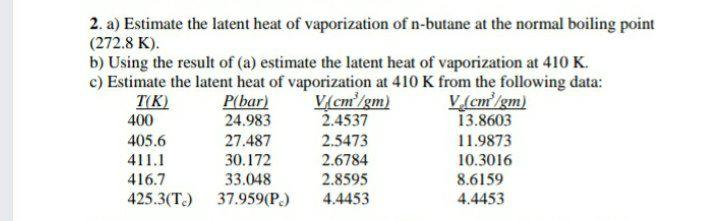
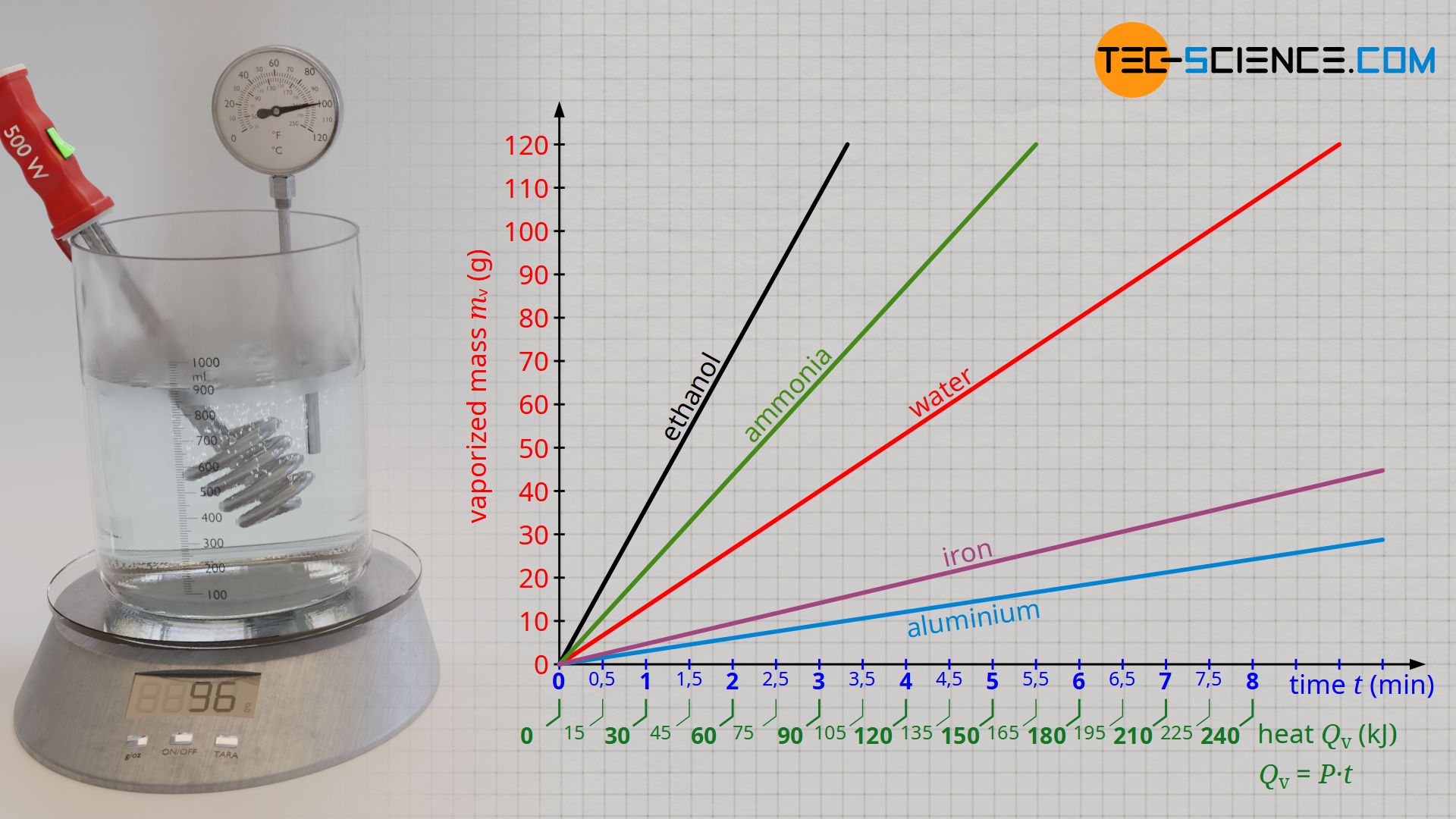
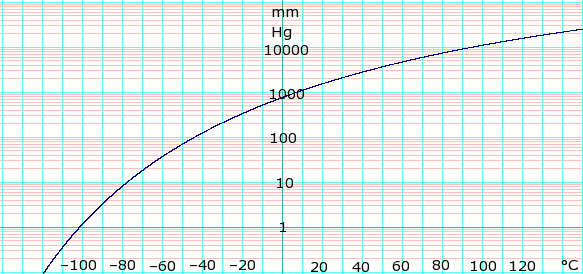
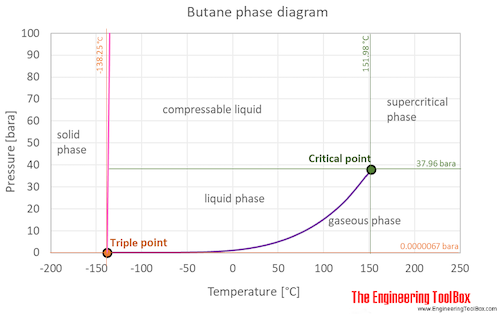
Approximated latent heat of vaporization for (a) butane; (b) isobutane;... | Download Scientific Diagram

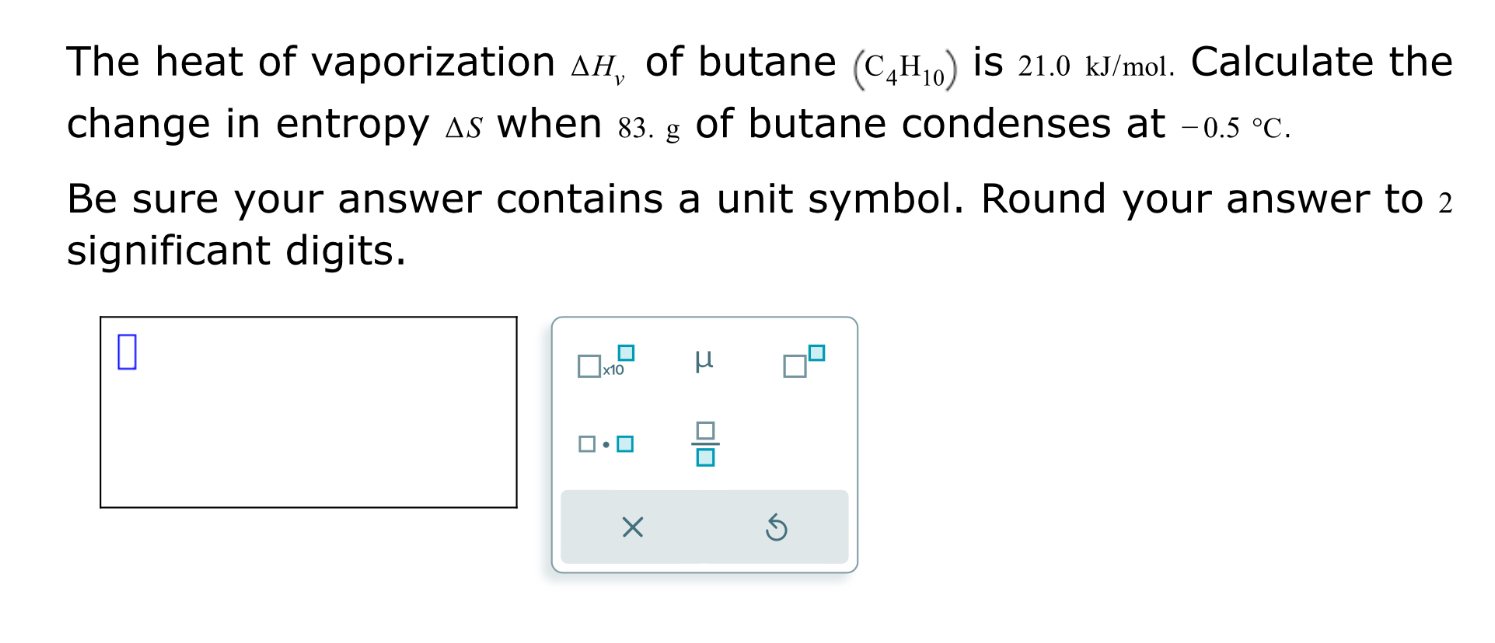
SOLVED: The heat of vaporization ΔHv of butane (C4H10) is 21.0 kJ/mol. Calculate the change in entropy ΔS when 60 g of butane condenses at -0.5°C. Be sure your answer contains a

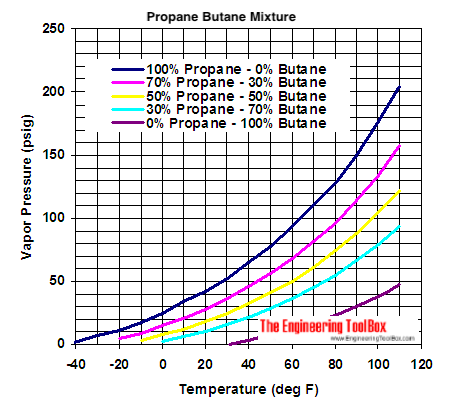
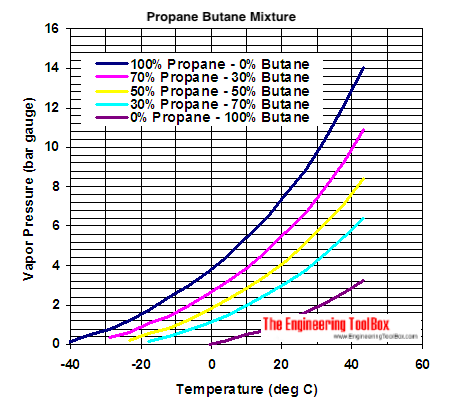

![Latent heat of vaporization for main components of LNG [10]. | Download Table Latent heat of vaporization for main components of LNG [10]. | Download Table](https://www.researchgate.net/publication/330572654/figure/tbl3/AS:718422421803010@1548296661881/Latent-heat-of-vaporization-for-main-components-of-LNG-10.png)